
It forces us to change our thinking in order to find it." "Everything we call real is made of things that cannot be regarded as real.“ “No, no, you're not thinking you're just being logical. A real life example would be a neon light, when the neon gas has electricity added to it the electrons shift around causing atoms to move which gives off the neon light.ġ4 Neils Bohr Quotes "Never express yourself more clearly than you are able to think.“ "Every great and deep difficulty bears in itself its own solution. For example when an electron goes from a low energy orbit such as n=1 to n=7 the atoms that have been moved by an electric discharge can give off light when the electrons move to the higher energy orbit. The light comes from the electric discharge that is created by the atoms that have moved. The electron can move from a low energy orbit to a high energy orbit and give off light or the electron can move from a high energy orbit to a low energy orbit and give off light. This Bohr model, a quantum model shows that hydrogen absorbs light when an electron moves from one energy level to another. Will Westenberger Jhace Smith Tiphani Cohens Samantha Silva-Nashġ3 Analysis and Conclusion From His Experiment The orbits are labeled by the quantum number n.ġ2 Neils Bohr: Contributions to the Atomic Theory He concluded that because of this quantization, electron orbits have fixed sizes and energies. He hypothesized that the angular momentum of the electron is quantized (which means it only has discrete values). Bohr composed an accurate formula for the energy levels of a hydrogen atom using Planck’s constant. Using the Young’s slit device, Bohr delimited two quantum paths between the source and the detecting plane. Which explains the structure and complexity in the actions of atoms. Later other physicists expanded his theory into quantum mechanics. 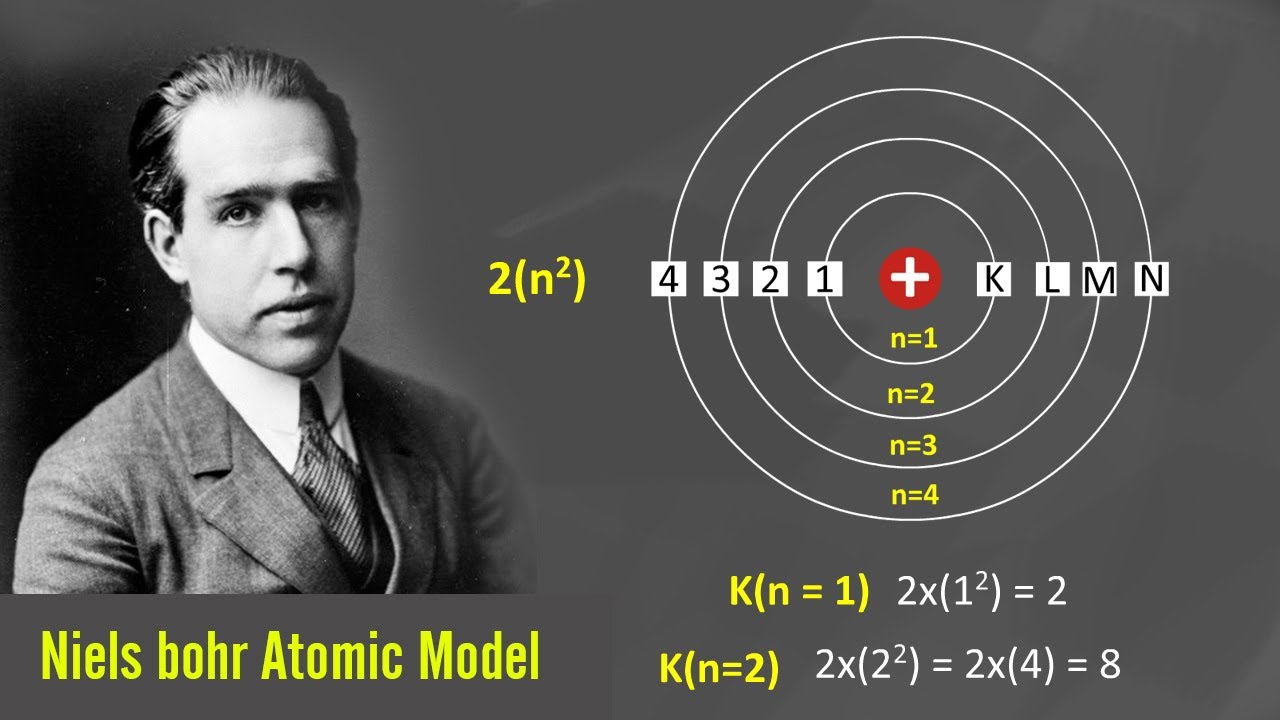
He also concluded that these outer orbits can determine an atom's chemical properties, and that Atoms emit radiation and light when an electron jumps from an outer orbit to an inner one. Proposed that Atoms moved In fixed paths around the nucleus, like planets orbiting a sun.īohr concluded that the outer orbits could hold more electrons than the inner ones. Electrons orbit around the nucleus which contains the Neutrons and Protons. The Bohr model gave way to the theory of Atoms, which is called Quantum mechanics. 1962 Returned to Copenhagen and furthered studied nuclear energy.ĩ The Bohr Model The Bohr Model of the atom revealed some of the most important properties of molecular and Atomic structure. Work on nuclear fission and created liquid droplet theory which described the way neutrons and protons behaved.ħ 1950’s In 1954, Neil assembled the Atoms for Peace Conference of In 1957, Bohr received the Atoms for Peace Award for his theories and examples to show how to use atomic energy responsibly. Quantum theory showed the specific relation proportion of an electron and how it circulates the atoms’ nucleusĥ 1922 Bohr received a Nobel Prize in Physics for work of "for his services in the investigation of the structure of atoms and of the radiation emanating from them." He continued to study new quantum principles which ultimately explained why light can be seen as a particle and wave but not at the same time He developed an explanation for atomic structure that uses the periodic table of elements.Ħ 1930 Neil clarified problems in quantum physics Also showed the light quantum of distinct energy, which formed the bases for the quantum theory. The model also purposed that electrons could drop from higher energy orbit to a lower orbit. This model proposed that electrons circled the atom’s nucleus in a certain way, therefore it also explained that electron traveled in specific energy. By the summer of 1920 it did not look very much like the atom introduced seven years earlier. 
Physicist, scientist, humanitarian, and mathematician Born OctoFrom Copenhagen, Denmark Died NovemFather was celebrated physicist Studied at University of Copenhagen Won 1922 Nobel Peace Prize in physicsģ Neils Bohr’s contribution to the Atomic Theory The contributions made by the German physicists, and to a lesser extent by Bohr himself, not only changed the national setting of atomic theory, they also considerably changed the picture of the Bohr atom. Where \( \Re \) is the Rydberg constant, h is Planck’s constant, c is the speed of light, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus.Presentation on theme: "Niels Henrik David Bohr"- Presentation transcript:
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
